Calculate the percentage by mass of urea in the solution. Jan 2008 - 38 What is the concentration of O 2(g), in parts per million, in a solution that contains 0.008 gram of O 2(g) dissolved in 1000.6 g of urea was dissolved in 500 g of water. grams of a solution having a concentration of 5 parts per million? June 2009- 36 What is the total mass of solute in 1000. June 2008- 38 What is the concentration of O 2(g), in parts per million, in a solution that contains 0.008 gram of O 2(g) dissolved in 1000. June 2005 -42 What is the concentration of a solution, in parts per million, if 0.02 gram of Na 3PO 4 is dissolved in 1000 grams of water? grams of H 2O, what is the concentration of the resulting solution, in parts per million? If 0.025 gram of Pb(NO 3) 2 is dissolved in 100. Here was another one, but it was a multiple choice. It is technically wrong not to add the solute and solvents together to determine the mass of solution. Even though mathematically it didn't change a thing. Īnswer-The equation above reads "grams of solution", you had to add the solute to the solvent in the denominator. Your response must include both a correct numerical setup and the calculated result. In the space in your answer booklet, calculate the dissolved oxygen concentration of this solution in parts per million. 
An aqueous solution has 0.0070 gram of oxygen dissolved in 1000. Most students lost 1 of the 2 points on this one.Ħ6. Then June 2007, New York State threw this curveball. What is the concentration of a solution, in parts per million, if 0.02 gram of NaCl is dissolved in 1000. This is really just a plug and chug problem. Think about this 1ppm = 1 inch in 16 miles  
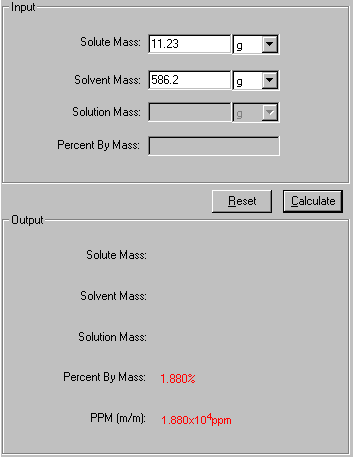
This is used for very small concentrations of particles in solutions. This is another way of determining concentration. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2023
Categories |
 RSS Feed
RSS Feed
